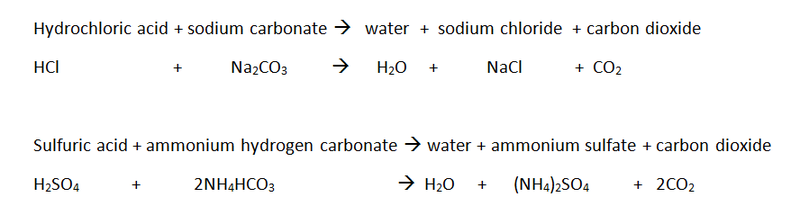Click here for some great helpful notes on Chemistry basics - atoms, ions, electron arrangement.......and more...check it out !!!
periodic table
noun CHEMISTRY
noun CHEMISTRY
- a table of the chemical elements arranged in order of atomic number, usually in rows, so that elements with similar atomic structure (and hence similar chemical properties) appear in vertical columns.
NOTE - Isotopes are NO LONGER part of this standard so you can IGNORE these !!!
Acids and Bases:
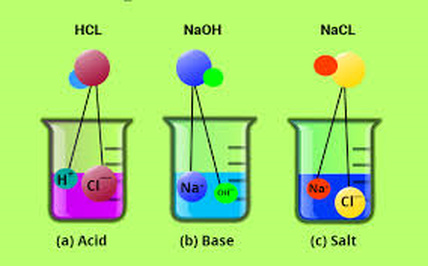
- Acids make up an important group of compounds that contain hydrogen. When acids dissolve in water, hydrogen ion (H+) is released into the resulting solution. They have a low pH - below 7
- A base is a substance that releases hydroxide ions (OH-) into solution. They have a high pH - above 7. Soluble bases are called ALKALI
- pH is a measure of the hydrogen ion concentration in a solution.
- An acid will dissociate in water producing hydrogen ions.ie: releases H + ions in solution
- A base (usually containing a metal) will dissociate in water to produce hydroxide ions. ie: releases OH- ions
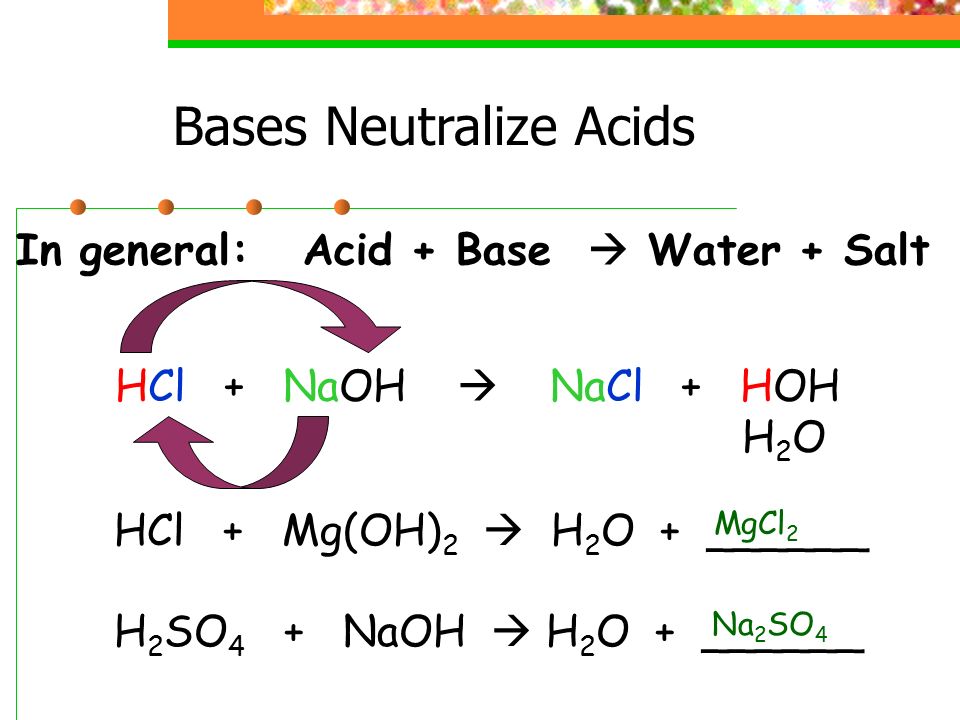
- Acids and bases will neutralize each other when mixed. They produce water and an ionic salt, neither of which are acidic or basic ie: a NEUTRAL solution is produced.

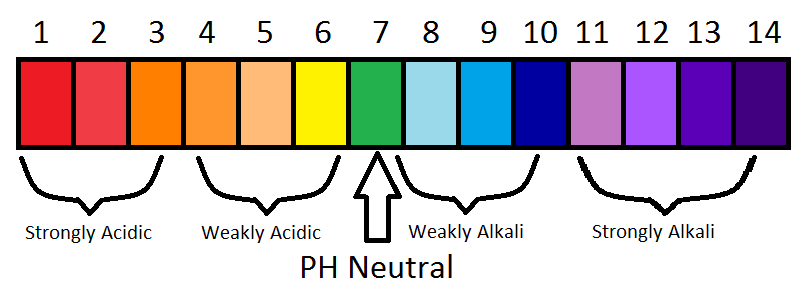
The pH Scale:
This is a scale for measuring how acidic or basic(alkaline) a solution is ! So is a measure of the Hydrogen Ion concentration in a solution.
Click here to check out a wee animation on Acids and Bases
//slideplayer.com/slide/9130506/
LEARN the first 3 of the common Acids and Bases above to achieve to your potential in the exams !!!!!!!!!!
You need to also know the way to MEASURE acidity levels using Indicators:
LITMUS is RED in Acidic solution and BLUE in alkaline solution
PHENOLPTHALEIN is COLOURLESS in ACIDIC solutions and PINK/RED in alkaline
UNIVERSAL INDICATOR has a "range" of colours depending on the pH of a solution eg: Red if pH 1, Green if pH 7 and purple if pH 14 (see pH scale)
You need to also know the way to MEASURE acidity levels using Indicators:
LITMUS is RED in Acidic solution and BLUE in alkaline solution
PHENOLPTHALEIN is COLOURLESS in ACIDIC solutions and PINK/RED in alkaline
UNIVERSAL INDICATOR has a "range" of colours depending on the pH of a solution eg: Red if pH 1, Green if pH 7 and purple if pH 14 (see pH scale)
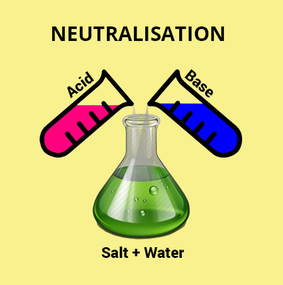
NEUTRALISATION:
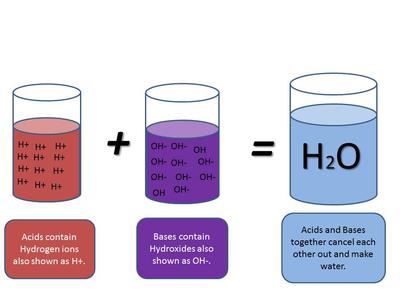
This is the reaction between an acid and a base - when they are in equal concentrations the base cancels out the effects of the acid (or vice versa) and the products are NEUTRAL.
Because an alkali dissociates in solution ie: releases its OH - ions and an acid dissociates in solution and releases H+ ions these 2 dissociated ions combine/collide and react to form WATER ie: H2O a neutral product.
Metals we use are - HCL, H2SO4 and HNO3
Bases we use are - any Metal Oxides, Metal Hydroxides, Carbonates and Hydrogen Carbonates
The equation is written:
ACID + BASE = SALT + WATER
The acid used determines the end name of the salt.
Hydrochloric acid produces a salt that will be named ___________chloride
Sulfuric acid produces a salt that will be named ___________sulphate
Nitric acid produces a salt that will named ___________nitrate
When an acid is neutralised by a base or vice versa WATER and a SALT are produced.
The resulting salt can be proven by evaporating the resulting liquid off and so that "salt crystals" are produced/left over.
Using neutralisation
Click on the Button below for a great website on acids and bases !!!
This is the reaction between an acid and a base - when they are in equal concentrations the base cancels out the effects of the acid (or vice versa) and the products are NEUTRAL.
Because an alkali dissociates in solution ie: releases its OH - ions and an acid dissociates in solution and releases H+ ions these 2 dissociated ions combine/collide and react to form WATER ie: H2O a neutral product.
Metals we use are - HCL, H2SO4 and HNO3
Bases we use are - any Metal Oxides, Metal Hydroxides, Carbonates and Hydrogen Carbonates
The equation is written:
ACID + BASE = SALT + WATER
The acid used determines the end name of the salt.
Hydrochloric acid produces a salt that will be named ___________chloride
Sulfuric acid produces a salt that will be named ___________sulphate
Nitric acid produces a salt that will named ___________nitrate
When an acid is neutralised by a base or vice versa WATER and a SALT are produced.
The resulting salt can be proven by evaporating the resulting liquid off and so that "salt crystals" are produced/left over.
Using neutralisation
- Farmers use lime (calcium oxide) to neutralise acid soils.
- Your stomach contains hydrochloric acid, and too much of this causes indigestion. Antacid tablets contain bases such as magnesium hydroxide and magnesium carbonate to neutralise the extra acid.
- Bee stings are acidic. They can be neutralised using baking powder, which contains sodium hydrogen carbonate.
Click on the Button below for a great website on acids and bases !!!
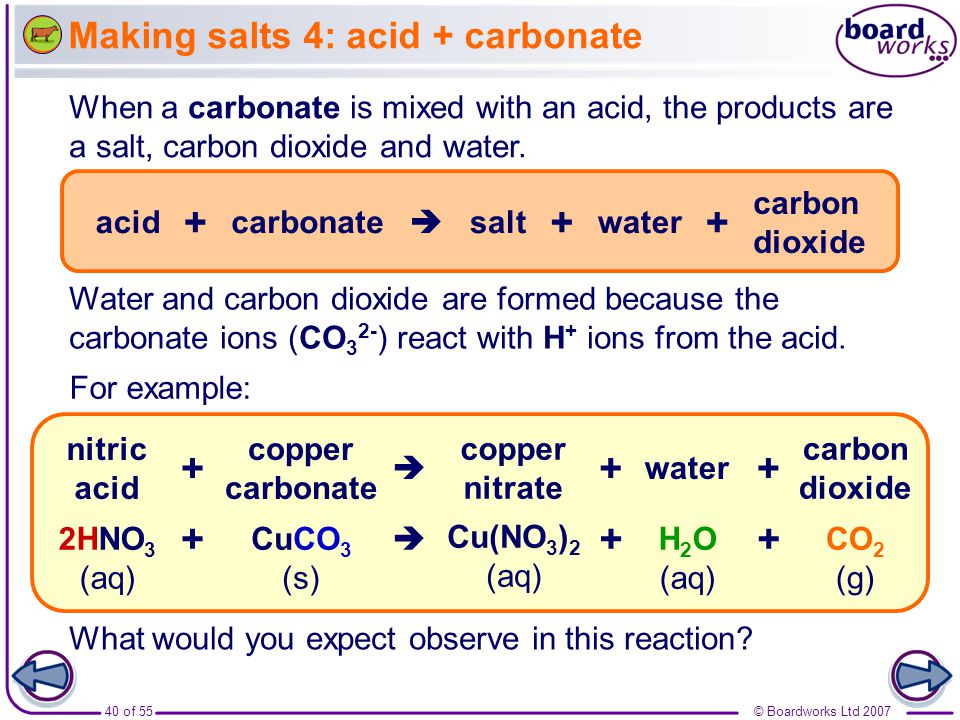
Carbonates and acids
Remember that Carbonates have the chemical symbol CO3
There are two main types of Carbonates:
Metal carbonates - these are made up of a metal and a carbonate ion - Eg: Calcium carbonate CaCO3 - Sodium Carbonate NaCO3
OR
Metal Hydrogen Carbonates - there are made up of a metal and a hydrogen carbonate ion eg: Sodium Hydrogen Carbonate NaHCO3 (commonly known as Baking Soda)
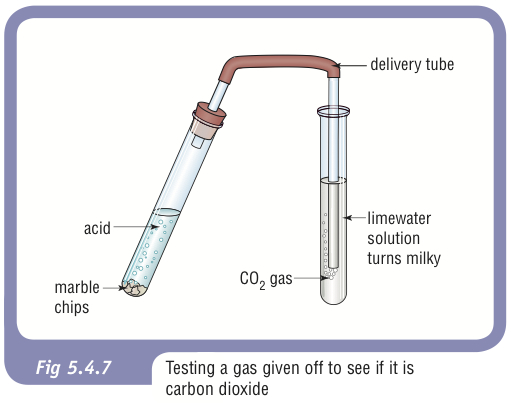
Whenever an acid and carbonate are reacted together Carbon Dioxide Gas is produced and can be observed by the Fizzing or bubbles produced in the reaction. You need to be able to know how to test for CO2 !!!
**** Test for CO2 Gas: Bubbling the gas through Limewater - if present Limewater wil turn milky white/cloudy *******
Remember that Carbonates have the chemical symbol CO3
There are two main types of Carbonates:
Metal carbonates - these are made up of a metal and a carbonate ion - Eg: Calcium carbonate CaCO3 - Sodium Carbonate NaCO3
OR
Metal Hydrogen Carbonates - there are made up of a metal and a hydrogen carbonate ion eg: Sodium Hydrogen Carbonate NaHCO3 (commonly known as Baking Soda)
Whenever an acid and carbonate are reacted together Carbon Dioxide Gas is produced and can be observed by the Fizzing or bubbles produced in the reaction. You need to be able to know how to test for CO2 !!!
**** Test for CO2 Gas: Bubbling the gas through Limewater - if present Limewater wil turn milky white/cloudy *******
WORD and CHEMICAL equations: