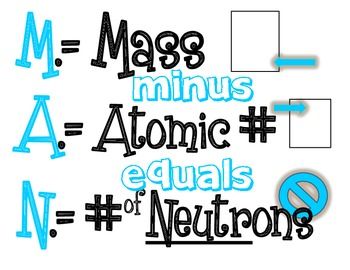WELCOME to EVERYDAY CHEMISTRY !!
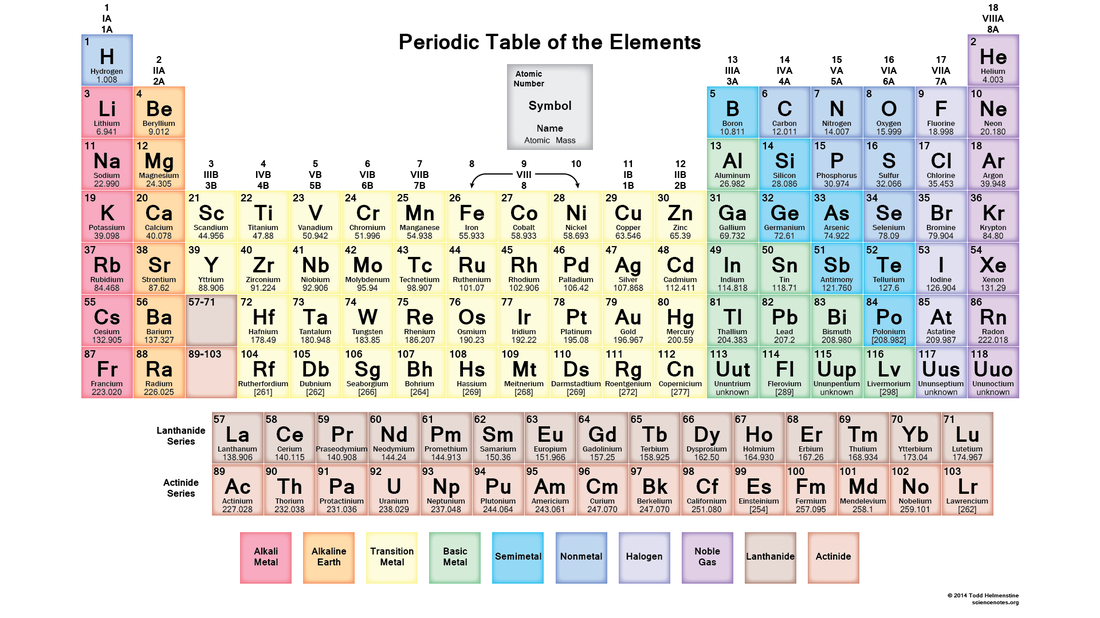
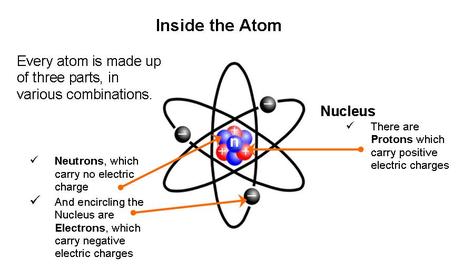
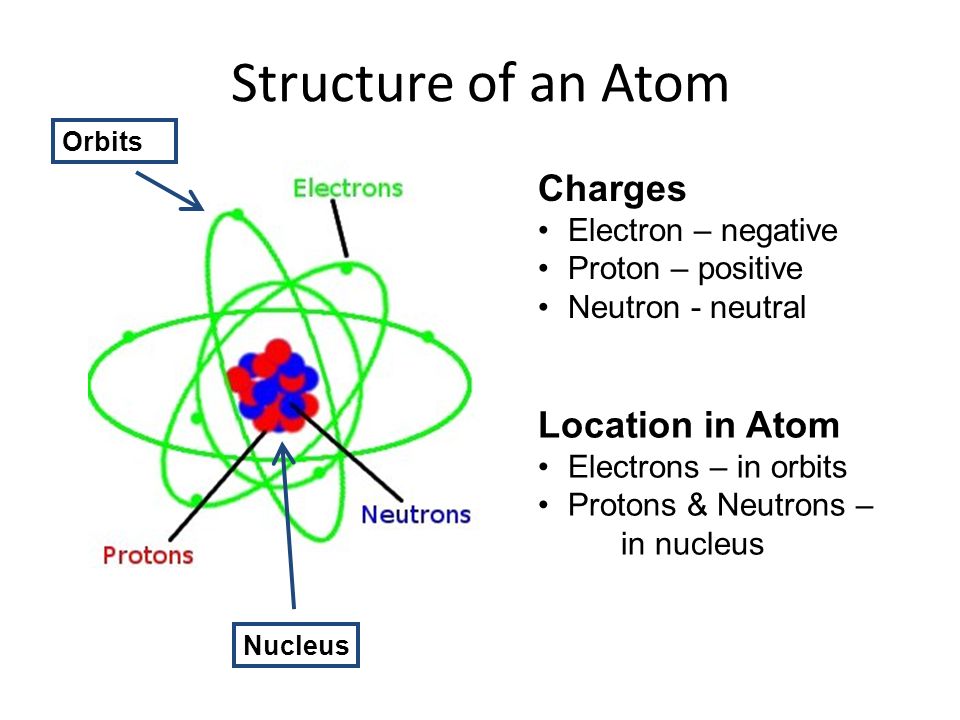
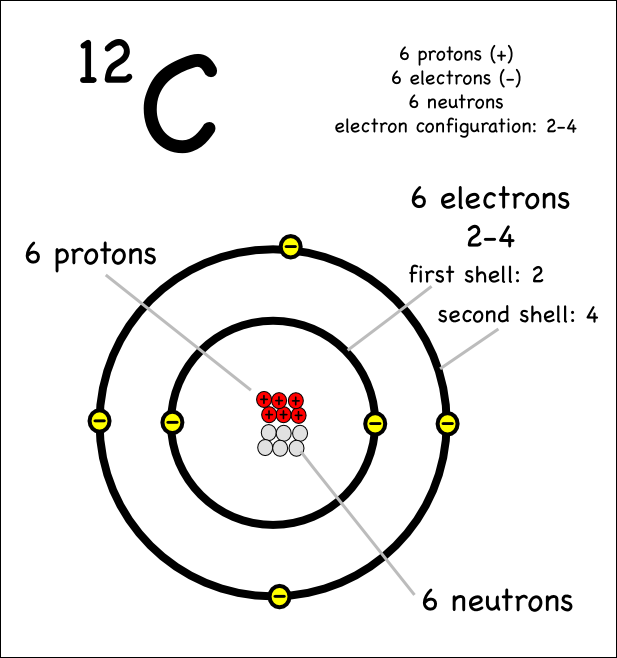
ATOMIC THEORY:
|
|
|
|
Check this out here....
|
Acids and Bases:
ACIDS
An acid is a sour tasting substance which neutralises a base and has a pH of less than 7. All acids contain Hydrogen and release its H + ions in water.
Acids frequently feel "sticky" to touch and are mostly liquids
**********ACIDS will turn Blue Litmus RED**********
Acids in the lab:
Dilute acids
You will have used some dilute acids at school, such as:
- Hydrochloric acid, HCl
- Sulphuric acid H2SO4
- Nitric acid. HNO3
Their bottles are labelled with the warning symbol for 'irritant'. This means that if any of them makes contact with your skin, it will become red or blistered. You must wash off any spills with plenty of water, otherwise your skin will soon feel as if it is burning.
Concentrated acids
You are unlikely to have used concentrated acids but your teacher might have shown you some experiments with them. This is because concentrated acids are corrosive. They can attack metals and destroy skin if spilled.
Acids in the home
Laboratory acids are far too dangerous to taste, but you will have swallowed some dilute weak acids. Acids have a sour taste, like vinegar, which contains ethanoic acid, and lemons, which contain citric acid. These are safe to use in food, but they can still hurt if they get into a cut or into your eyes.
Other acids you will find at home are carbonic acid in fizzy drinks,
tannic acid in tea and ascorbic acid which is vitamin C, found in fruit and vegetables.
BASES
A base is a soapy substance which neutralises an acid and has a pH of more than 7. It releases OH - ions (Hydroxide ions) into solution.
Bases are bitter to the taste (like Baking Soda) often used as cleaners - they have a slippery feel to touch and are liquids and solids.
********* BASES turn RED litmus BLUE*********
A base which is SOLUBLE in water is called an ALKALI and the solution it creates with water is called alkaline.
Alkali is often used instead of the word Base.
Common Lab Bases are:
Sodium Hydroxide NaOH
Potassium Hydroxide KOH
Ammonium Hydroxide NH4OH
Other bases we will work with are:
Calcium Carbonate CaCO3 (marble Chips)
Ammonia NH3
INDICATORS:
Indicators can be used to determine the pH of a solution by the colour change. (how acidic or basic the solutions is)
An indicator is a large organic molecule that works like a "colour dye". They respond to a change in the hydrogen ion concentration of a solution.
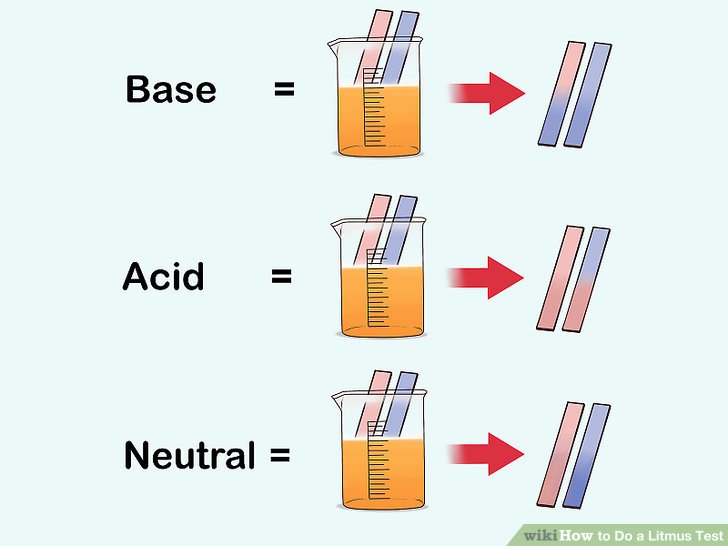
The most common indicator is found on litmus paper.
This is a special paper that can be used to tell us if something is an acid or an alkali (base)
Red Litmus remains red for acid and turns blue for a base.
Blue Litmus paper turns Red for an acid and remains blue in a base
Another type of indicator is Universal Indicator
Universal Indicator, which is a solution of a mixture of indicators that provide a full range of colours for the pH scale.
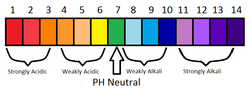
It indicates to us the "strength" of the acid or base. (see the diagram/pictures below)
It shows Neutral as GREEN and then a colour range for strong and weak acids and strong and weak bases !!
A pH Scale can be used to give us the exact STRENGTH of an acid or Alkali - we use this along side our Universal Indicator.
Indicators can be used to determine the pH of a solution by the colour change. (how acidic or basic the solutions is)
An indicator is a large organic molecule that works like a "colour dye". They respond to a change in the hydrogen ion concentration of a solution.
The most common indicator is found on litmus paper.
This is a special paper that can be used to tell us if something is an acid or an alkali (base)
Red Litmus remains red for acid and turns blue for a base.
Blue Litmus paper turns Red for an acid and remains blue in a base
Another type of indicator is Universal Indicator
Universal Indicator, which is a solution of a mixture of indicators that provide a full range of colours for the pH scale.
It indicates to us the "strength" of the acid or base. (see the diagram/pictures below)
It shows Neutral as GREEN and then a colour range for strong and weak acids and strong and weak bases !!
A pH Scale can be used to give us the exact STRENGTH of an acid or Alkali - we use this along side our Universal Indicator.

ALL things Acids and Bases...check this out to enhance your understanding of Acids and Bases.....
Brave enough for some questions...have a go at this quiz....
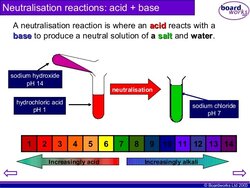
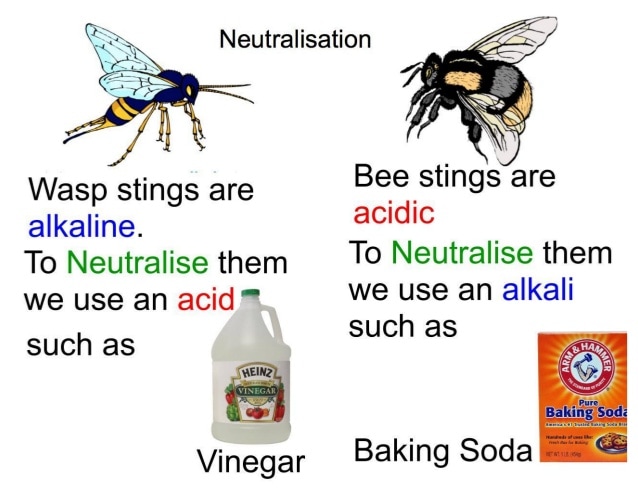
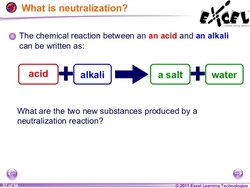
NEUTRALISATION:
This is the reaction between an acid and a base.
When they are in equal concentrations the base cancels out the effects of the acid (or vice versa) and the products are NEUTRAL.
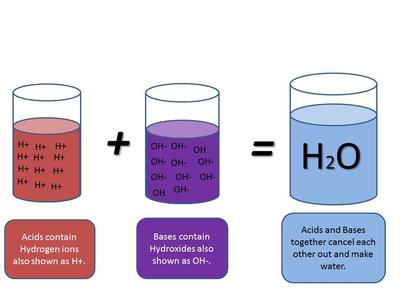
Because an alkali dissociates in solution ie: releases its OH - ions and an acid dissociates in solution and releases H+ ions these 2 dissociated ions combine/collide and react to form WATER ie: H2O a neutral product. Hence a NEUTRAL solution is created.
This is the reaction between an acid and a base.
When they are in equal concentrations the base cancels out the effects of the acid (or vice versa) and the products are NEUTRAL.
Because an alkali dissociates in solution ie: releases its OH - ions and an acid dissociates in solution and releases H+ ions these 2 dissociated ions combine/collide and react to form WATER ie: H2O a neutral product. Hence a NEUTRAL solution is created.
Indigestion and Acid Reflux
In our stomach we produce acid to help break down food into smaller molecules. Acid Reflux can happen because we are producing too much acid in our stomach. This starts to come up the oesophagus (pipe leading from stomach to the mouth) and causes a burning sensation that is very uncomfortable. Swallowing a base like baking soda, produces CO2 (makes us burp), salt and water. This is neutral and has a ph of 7 and the burning stops.
In our stomach we produce acid to help break down food into smaller molecules. Acid Reflux can happen because we are producing too much acid in our stomach. This starts to come up the oesophagus (pipe leading from stomach to the mouth) and causes a burning sensation that is very uncomfortable. Swallowing a base like baking soda, produces CO2 (makes us burp), salt and water. This is neutral and has a ph of 7 and the burning stops.